
Trapping carbon with iron
Iron fertilising of the oceans can make plankton draw more carbon dioxide down to the seabed for long-term storage.
Denne artikkelen er over ti år gammel og kan inneholde utdatert informasjon.
Trillions of miniscule algae in seawater are constantly removing CO2 from the atmosphere. Such algae blooms consist of unicellular phytoplankton that break down molecules of the climate gas through photosynthesis, releasing oxygen. When they die, many of them sink to the seabed, along with the carbon they have assimilated.
Centuries can pass before this carbon regains contact with the atmosphere.
A number of factors combine to determine the quantity of carbon drawn into the ocean depths by means of this biological pump.
Fertilising algae blooms with iron has been suggested as a form of geo-engineering for years. Now a team of Norwegian and international researchers have conducted an experiment that could possibly be an effective way to mitigate some of the effects of our greenhouse gas excesses.
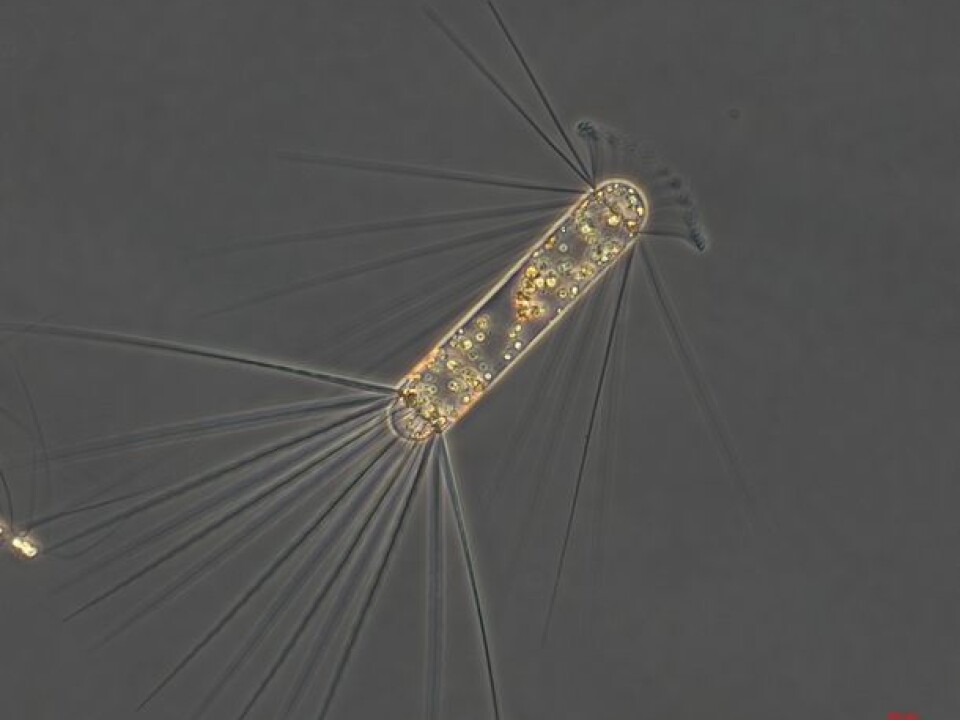
Their experiments from the Southern Ocean show a good sprinkling of the metal speeds up the growth of the CO2 consuming plankton and a large share of them sink to the sea floor when they die.
This was the first large-scale study showing that many of the tiny plankton from a bloom fertilised with iron actually disappear into the depths, according to Richard Bellerby of the Norwegian Institute for Water Research (NIVA). The study is published today, in Nature.
Iron deficiency
The research backs up the so-called iron hypothesis, initially proposed by the American oceanographer John Martin in the late 1980s to explain one of the mysteries of the seas:
In certain ocean regions, for instance the Southern (Antarctic) Ocean and some of the North Pacific Ocean, nutrients such as nitrogen and phosphorus are plentiful. The sun delivers sufficient light for algae growth. Phytoplankton is present in these waters, but the organisms don’t flourish to the point of becoming blooms.

What’s stopping them?
Martin’s idea was that algae need small amounts of trace elements, especially iron, in addition to various nutrients. In seawater containing little iron, the plant plankton just don’t thrive.
Iron fertiliser
Experiments have shown since the early 1990s that phytoplankton blooms can be started by fertilising the ocean with iron. But the next question is what happens to all these tiny plant-like organisms in a bloom?
If many of them sink to the bottom and remain there in the depths, the result could be that carbon is sequestered for centuries. But scientists haven’t been able to ascertain this.
So a team of international researchers, including Richard Bellerby from NIVA/The Bjerknes Centre for Climate Research and Philipp Assmy from the Norwegian Polar Institute, have conducted a number of experiments to literally get to the bottom of the issue.
They’ve managed to follow the fate of diatoms − a major group of algae and one of the most common types of phytoplankton − after releasing iron into Southern Ocean eddies.
Fertilised vortices
Along the wavy edge of major ocean currents, large spiralling whirlpools or eddies are formed. The biggest of these can last for weeks or months.
During this period, the core of the vortex forms an isolated column of water stretching from the ocean surface to the dark depths of the sea. With such an unbroken column of water at their service, the researchers were able to follow the tiny algae down through the saltwater strata.
A diatom bloom cropped up four weeks after the iron fertilisation in the nutrient-rich waters. Most of these tiny algae died soon afterwards and began to sink through the depths.
Deep, deep down
The researchers observed sheets of slime consisting of dead cells sinking rapidly. When all their measurements were assessed these indicated at least half of the algae from the bloom sank deeper than 1,000 metres.
The scientists in the Nature article that it’s likely a large share of them ended up on the seabed.
Bellerby informs us this is the first study to show large-scale transport of carbon away from the surface waters.
Once carbon reaches the murky depths, it takes a long time before it comes in contact with the surface again.
Not a climate tool yet
“The deeper the organic carbon sinks, the longer it will be isolated from atmospheric contact,” says Assmy.
The carbon that sinks further down than a kilometre can be sequestered for centuries and even longer if it gets packed under pressure into the seafloor sediments and lithified into rock.
Nevertheless, Bellerby and Assmy warn against viewing these results as proof that we can use iron fertilising as an initiative against the increasing CO2 levels in the atmosphere and resulting climate changes.
Bellerby stresses that as of now nobody can steer the processes that are involved in the biological pump.
Before humankind can consider any practical use of these findings, much more understanding is needed about the complex physical, ecological, biochemical and climatic couplings in the Antarctic Ocean.
“Iron fertilising can be one of several methods to reduce the CO2 in the atmosphere, but it will never be a solution to global warming,” says Assmy.
Reference:
V. Smetacek et al., Deep carbon export from a Southern Ocean iron-fertilized diatom bloom, Nature, 19 July 2012, vol. 487, pp. 313 – 319.
---------------------------
Read the story in Norwegian at forskning.no
Translated by: Glenn Ostling


































